#COVID #treatment #ivermectin
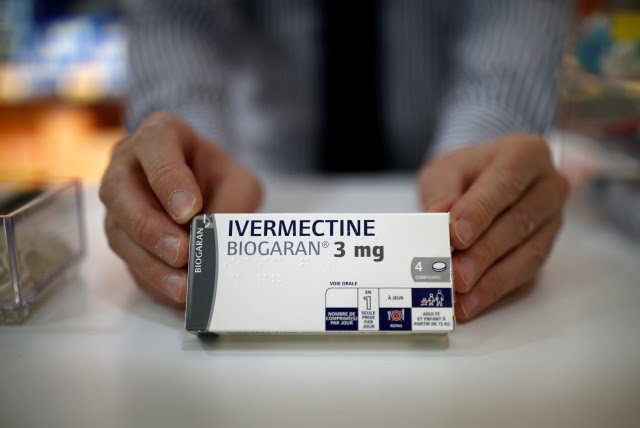
A double-blind study of an antiparasitic drug that’s been in the news a lot lately not only shows that the drug can reduce the duration and infectiousness of COVID-19, but can do all that on less than $1 a day.
That drug is ivermectin, and Professor Eli Schwartz, founder of the Center for Travel Medicine and Tropical Disease at Sheba, says his studies, recently published on RXiv, show it is a cost-effective treatment that can help shorten the infection period. Dr. Schwartz studied 89 volunteers diagnosed with the virus, 50% received ivermectin; the other 50%, a placebo.
“Nearly 72% of volunteers treated with ivermectin tested negative for the virus by Day 6,” The Jerusalem Post reported. “In contrast, only 50% of those who received the placebo tested negative.” And, besides feeling better quicker, fewer ivermectin patients also were found to be infectious: 13%, compared to 50% of the placebo group.
The findings also showed that “there is almost a 100% chance that a person will be noninfectious in four to 6 days,” Dr. Schwartz said.
Ivermectin is an inexpensive drug that was approved by the US FDA in Y 1987 for treatment of a parasitic roundworm.
The loudest opposition to ivermectin has come from Merck Co (NYSE:MRK), which manufactured the drug in the 1980s.
In a public statement about ivermectin on its website in February, it said: “Company scientists continue to carefully examine the findings of all available and emerging studies of ivermectin for the treatment of COVID-19 for evidence of efficacy and safety. It is important to note that, to date, our analysis has identified no scientific basis for a potential therapeutic effect against COVID-19 from pre-clinical studies; no meaningful evidence for clinical activity or clinical efficacy in patients with COVID-19 disease, and a concerning lack of safety data in the majority of studies.”
Merck has not launched any studies of its own on ivermectin.
Have a healthy, prosperous day, Keep the Faith!